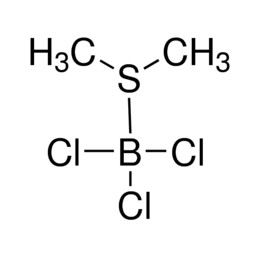
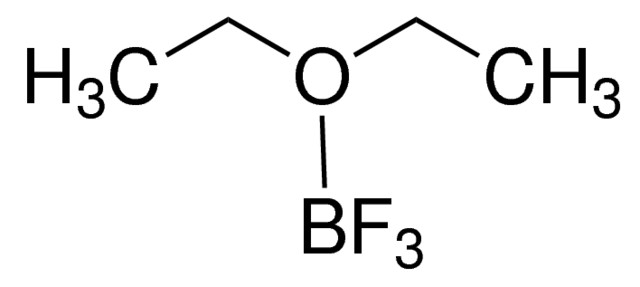
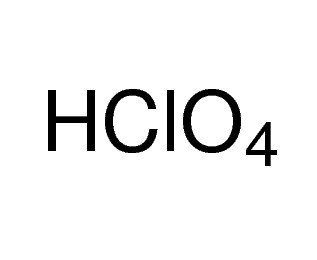
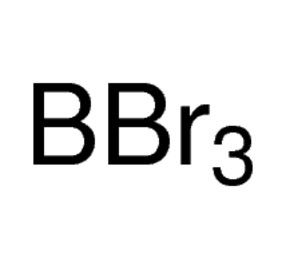Perchloric acid 70%, 99.999% trace metals basis
Perchloric acid (HClO4) can be used in the preparation of:
• Silica supported perchloric acid (HClO4-SiO2) which is used as a catalyst in geminal diacylation of aldehydes, synthesis of amidoalkyl naphthols,
primary carbamates, and tetrasubstituted imidazoles.[3][4][5]
[6]
• Alumina supported perchloric acid (Al2O3-HClO4), which is used as a catalyst in the synthesis of
• Fe3O4@SiO2-HClO4 catalyst, which is used in the synthesis of 1,4-dihydropyridine derivatives.
[8]
Perchloric acid can be used as:
• A reaction medium in oxidation reactions.[9][10]
•
A reagent for the preparation of fluorinated polymer electrolytes using poly (vinylidenefluoride-cohex
•
A dopant in the preparation of perchloric acid-functionalized polyaniline (HClO4/PANI) catalyst for the synthesis of 2-substituted benzothiazoles by condensation reaction between o-aminothiophenol with aldehydes.[12]
1 L in glass bottle
50, 250 mL in glass bottle